By: Isabela Claret Torres, MSc, Ph.D.
Biodegradation is a term commonly used when discussing environmental issues. And even though everyone has study this in school few remember the real steps of the process and what really means. Biodegradation in simple words is the decomposition of the dead organic matter by the biological community. It is a beautiful natural process that needs the cooperation of the whole microbial community. It is another way of nature telling us that we all have to cooperate in life.
What Does Biodegradable Mean?
When we say something is biodegradable it means it passes through the process of being reduce to small or soluble constituents by the biological community. The perfect biodegradation produces only water and carbon dioxide. And although we want every residue that we produce to be biodegradable, it is not always the best choice. Some substances when degrade, can generate more toxic intermediates than the original one. Biodegradation occurs in all types of habitats, natural or manmade that have microorganisms: on water, soil, lakes sediments, landfills, composting boxes, sewage treatment, you name it.
Organic Matter
All organic material that dies goes through decomposition. The first step is the break down of the big particles into small pieces that is done by insects, earthworms, or other organisms. This is concomitant or followed by the break down of the particulate matter into the dissolved phase. This is done by the production of extracellular enzymes. Extracellular enzymes, as the name says, are present outside of the microorganisms’ cells. It can be attached to the cell or loose in the surrounding environment. In most cases, an enzyme is a protein that is capable of breaking down organics into inorganics and soluble forms.

This is the way that the microbial community acquires inorganic nutrients and dissolved carbon. Enzymes are specific for the substrate they degrade. As an example, there is the Phosphatase that are specific to degrade different types of organic phosphorus. Or the Glucosidase that breaks down the cellulose into glucose, where it can be taken up by the microorganism. Many types of microorganisms produce extracellular enzymes used in the degradation process.
Biomass
The soluble substances can now be taken up by the microorganism cells and be used to build biomass and for other cellular function, as respiration. The complete degradation of the glucose is described by the following formula, where we can see, that glucose is totally reduced to CO2 and H2O:
Many different microorganisms participate in the biodegradation process. The aerobic bacteria, fermenters, nitrate reducers, iron and manganese reducers, sulfate reducers and methanogens. This last one’s strict anaerobic microorganisms. An aerobic environment is when there is oxygen, and aerobic bacteria can only live and be active when oxygen is present. While anaerobic is where there is no oxygen, and for anaerobic bacteria oxygen is very toxic.
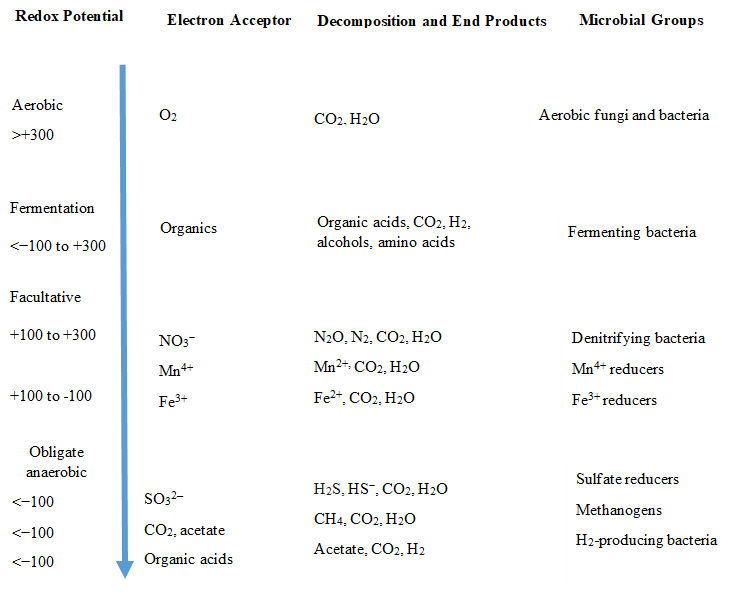
Figure 1. General scheme of the degradation or organic matter and its end products. (Source: Reddy and DeLaune 2008).
The Fig.1 is a scheme showing how organic matter is degraded. To be very simple, the Redox Potential measures the quantity or presence of oxygen. High positive numbers indicate aerobic environments, and very negative numbers completely anaerobic habitats. In each of these habitats completely different microbial communities are present.
Aerobic Microorganisms
The electron acceptor is involved in the energy production. Aerobic microorganisms are more energetically efficient than all the others, because the complete degradation with oxygen produces more energy than any other type. Consequently, when there is oxygen and high redox potentials the aerobic degradation of organic matter dominates. When oxygen became scarce the fermentative and facultative bacteria become active in the degradation process. Facultative organisms can live in either habitat, with or without oxygen. And finally, just when redox potentials are very low and there is no trace of oxygen the obligate anaerobes are present and functioning. Temperature, humidity, pH all can affect the decomposition process.
Compost
One curiosity of the decomposition process is about the smell that can produce. Most people think that the bad smell comes from the methane (CH4) production but is not. Methane is an odorless gas, but highly flammable. The smell of rotten eggs that comes from the sulfate reducers that can produce hydrogen sulfide (H2S). Fermentative products can also smell bad. In lakes sediments or wetlands, soils you can see a clear gradient of redox potential and of the microbial community involved in the degradation of organic matter or compost.
The beautiful thing about the biodegradation process is that each microbial group contributes and depends on each other to complete the process. In each step wastes are produced, as gases or dissolved compounds. When build up in the environment this end products becomes toxic to the microbial group that produced them. The next group uses the end product of the upper one and make it possible for them to coexist depending in each other, helping each other. Each one do its part, do a little and the organic matter is totally degraded. This is called syntrophy (Fig. 2).
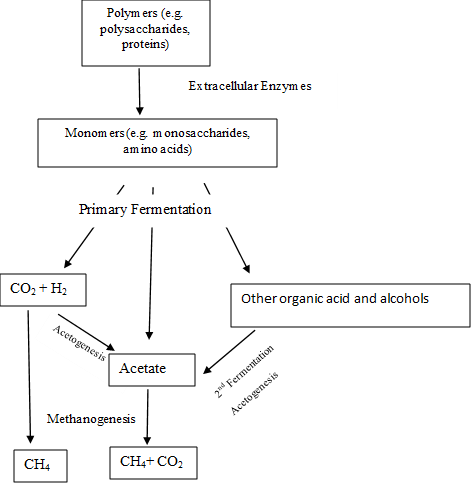
Figure 2. Metabolic scheme for the degradation of complex organic matter with syntrophic relationships among the microbial community. Source: Schlesinger 2005.
Carbon Footprint
That’s the lesson nature wants to give us. We can all do a little and help each other and make something big together. To help the environment we can start with small changes. One is to reduce our garbage. Keep always the 3 R’s rule in your mind: Reduce, Reuse and Recycle. Other ways include having a composting box at home or by offsetting your carbon footprint. Doing a little, one helping another to make big changes to protect our planet! So let’s start with small steps!
About the Author:
Isabela started her work with the environment in 1993 as a Biology Bachelor student at the highly regarded Federal University of Minas Gerais (UFMG/Brazil). Later she joined the Graduate Program on Ecology, Conservation and Management of the Wildlife of the same University (UFMG/Brazil) where she received her Masters degree in 1999. Later, she received her Ph.D. on Soil and Water Science at the University of Florida (USA). During her Ph.D. studies she received an Outstanding Student Poster Award at the ASLO (American Society of Limnology and Oceanography) 2006 Summer Meeting held in Victoria (Canada). After working for the State Government Secretary, consulting environmental company and watershed management company she began her post-doctorate in the Geography Department at UFMG (Brazil). Throughout her career she gathered knowledge and specialized on Biology, Ecology, Soil Science, Limnology, Paleolimnology, Biogeochemistry, Organic Geochemistry, Metal and Environmental pollution.
References:
Reddy, K.R. and DeLaune, R.D. 2008. Biogeochemistry of Wetlands: Science and Applications. CRC Press.
Schlesinger, W.H. 2005. Biogeochemistry. Treatise on Geochemistry Volume 8. Elsevier.

